Eumelanin contains a O-C-C-O semiquinone and gives an EPR signal consistent with an oxygen radical (a singlet). The former is associated with dark (brown or black) colors, the latter with red. This polymer is made of varying amounts of two monomers, called eumelanin and pheomelanin.
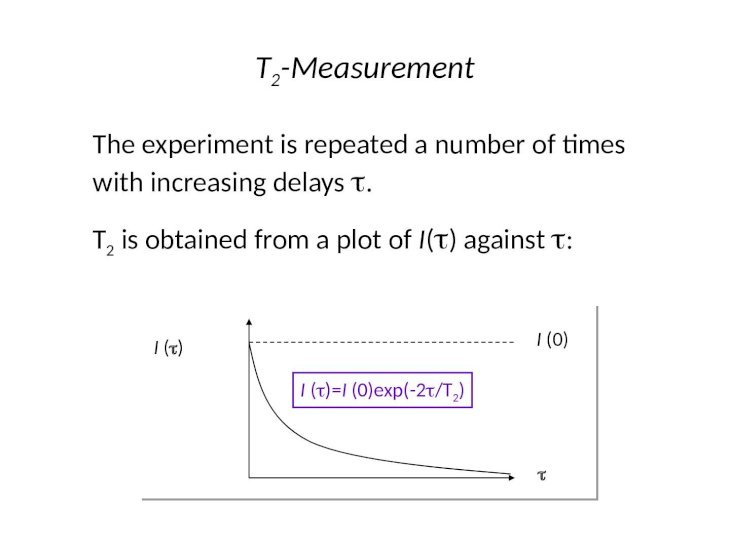
T1 MEASUREMENT NMR MESTRENOVA SKIN
Now that we are all experts on EPR, what is it that Chikvaidze and co-workers are measuring? There is a branched polymer in skin and hair called melanin that controls pigmentation color. Second, the y-axis is the first derivative of the absorbance. First, the x-axis is magnetic field (in gauss or telsa) not frequency. EPR spectra differ in two key ways from NMR spectra. (As an aside, I'll mention to organic chemists that they should look at the CDCl3 signal in a 1D 13C spectrum to see an equivalent effect). If the nuclei is 14N, the EPR signal is a triplet, but all three legs have identical height. Hence if the nuclei is 15N (I = 1/2) the EPR signal is a doublet. Although the g-factor is not diagnostic in these cases, the coupling to magnetic nuclei are! The unpaired electron will experience "hyperfine" coupling to nuclei, such as 14N (nuclear spin I = 1). For organic (oxygen, carbon and nitrogen) radicals, though, it seems like all g-factors are ~ 2. Of course it is! Spin-orbit coupling depends on which orbital the unpaired electron resides. I don't want to suggest that the spin-orbit coupling is not sensitive to electronic structure. With EPR, on the other hand, I am not aware of any standard tables of g-factors. These days every organic chemistry textbook contains a table of chemical shifts classified by functional group. In EPR the resonance frequency is reported as the "g-factor." Instead of depending on the shielding of nuclei by electrons like the chemical shift in NMR, the g-factor depends on the coupling of the spin motion of the electron to the orbital motion (spin-orbit coupling).
T1 MEASUREMENT NMR MESTRENOVA PLUS
In a simple 1D 1H NMR spectrum parameters like the resonance frequency (the chemical shift plus any scalar couplings) and peak integrals can be interpreted to understand properties of the molecule, for instance the molecular structure of a small drug-like organic molecule. To review, one can think about EPR much like you think about NMR. Tahoces, I.I discussed EPR briefly in an earlier post (). Javier Sardina (2004), Wavelet-based ultra-high compression of multidimensional NMR data sets, J. Tahoces, Manuel Martin-Pastor, Mónica Penedo, F. Having the ability to access directly to the processed spectrum without the need to reprocess it may be very handy. In this particular case, opening this single spectrum would take several seconds (if not minutes). Just to give a simple example: You have processed a 2D spectrum which was acquired with a NUS scheme and you have applied some additional time-consuming analysis operations (i.e. Unfortunately, this does not work well for many applications and introduce some additional difficulties. On the other hand and considering again the point that Mnova always keeps a copy of the original FID, why we don’t just save this FID plus the processing commands required to reconstruct the processed spectrum as other NMR applications do? Actually, this is a nice approach (under some circumstances) and would yield the best compression ratio achievable.
